Coexistence of vitreous and crystalline phases of H2O at ambient temperature
Understanding the phase behavior of H2O is essential in geoscience, extreme biology, biological imaging, chemistry, and physics. High-density amorphous (HDA) phase of H2O is of particular importance due to its extensive application in preserving biological samples for imaging at cryogenic temperatures. In this collaborative research project, the scientists confirmed the formation of ice HDA at room temperature while included grains of crystalline ice VI. Their study revealed that neither of the two common imaging techniques including: X-ray diffraction (XRD) and Raman accurately characterize the exact microstructure of the mixed phase, and thus a parallel characterization is required. This conclusion is supported with molecular dynamics (MD) simulations that is conducted by Ali K. Shargh, a Ph.D. student in ACMML laboratory at University of Rochester. Ali’s simulations confirm that Raman identifies amorphous while XRD captures the crystalline constituent of the mixed phase of H2O. The results have implications for experimental studies of biological systems as well as related metastable transitions in other materials under pressure.
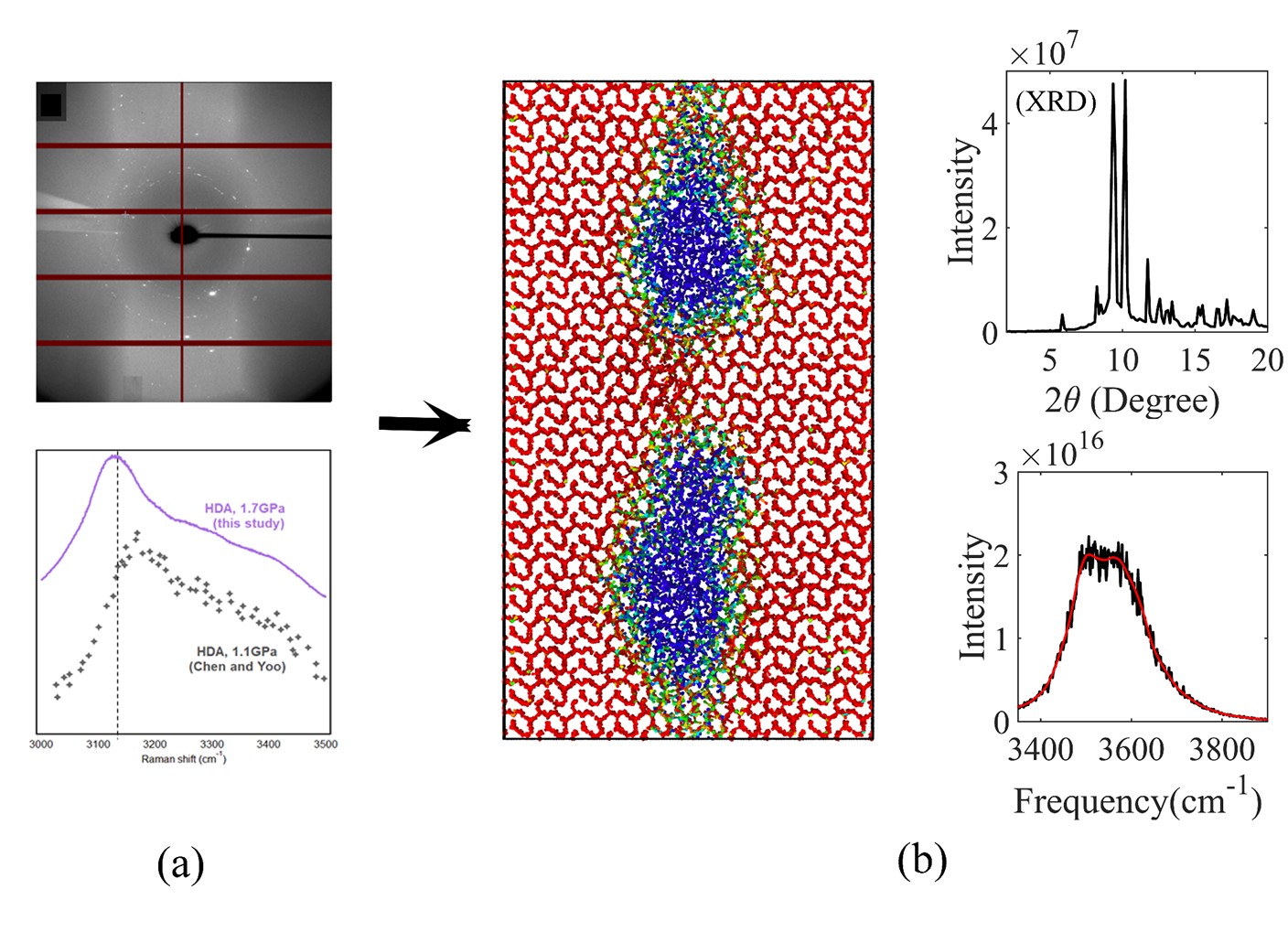
For more information, see: Ali K. Shargh, Aude Picard, Rostislav Hrubiak, Dongzhou Zhang, Russell J. Hemley, Shanti Deemyad, Niaz Abdolrahim, and Saveez Saffarian. (2022) Co-existence of vitreous and crystalline phases of H2O at ambient temperature
PNAS 119 (27) e2117281119

