Pressure enabling “molecular anvils” and modifying the energy landscape of chemical reactions
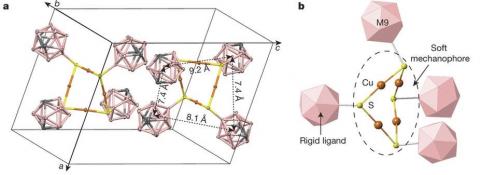
By engineering molecules with mechanically heterogeneous components with a compressible (‘soft’) mechanophore and incompressible (‘hard’) ligands, a research team has created ‘molecular anvils’, resulting in isotropic stress that leads to relative motions of the rigid ligands, anisotropically deforming the compressible mechanophore and activating bonds. Conversely, rigid ligands in steric contact impede relative motion, blocking reactivity. X-ray experiments, including the use of x-ray absorption measurements at HPCAT, demonstrate hydrostatic-pressure-driven redox reactions in metal–organic chalcogenides, incorporating molecular elements that have heterogeneous compressibility, in which bending of bond angles or shearing of adjacent chains activates the metal–chalcogen bonds, leading to the formation of the elemental metal. These results reveal an unexplored reaction mechanism and suggest possible strategies for high-specificity mechanosynthesis. More in H. Yan, et al, Nature, 554, 505-510, 2018.